 This expression does follow the convention Δ X = X(final) − X(initial) since −Δ E = −( E(final) − E(initial)) = E(initial) − E(final).Įquivalently, electron affinity can also be defined as the amount of energy required to detach an electron from the atom while it holds a single-excess-electron thus making the atom a negative ion, i.e. The usual expression for calculating E ea when an electron is attached isĮ ea = ( E initial − E final) attach = −Δ E(attach) Negative values typically arise for the capture of a second electron, but also for the nitrogen atom. In this case, the electron capture is an endothermic process and the relationship, E ea = −Δ E(attach) is still valid. However, if the value assigned to E ea is negative, the negative sign implies a reversal of direction, and energy is required to attach an electron. The relation between the two is E ea = −Δ E(attach). Confusion arises in mistaking E ea for a change in energy, Δ E, in which case the positive values listed in tables would be for an endo- not exo-thermic process. It is the word "released" within the definition "energy released" that supplies the negative sign to Δ E. The positive values that are listed in tables of E ea are amounts or magnitudes. Electron capture for almost all non- noble gas atoms involves the release of energy and thus are exothermic. 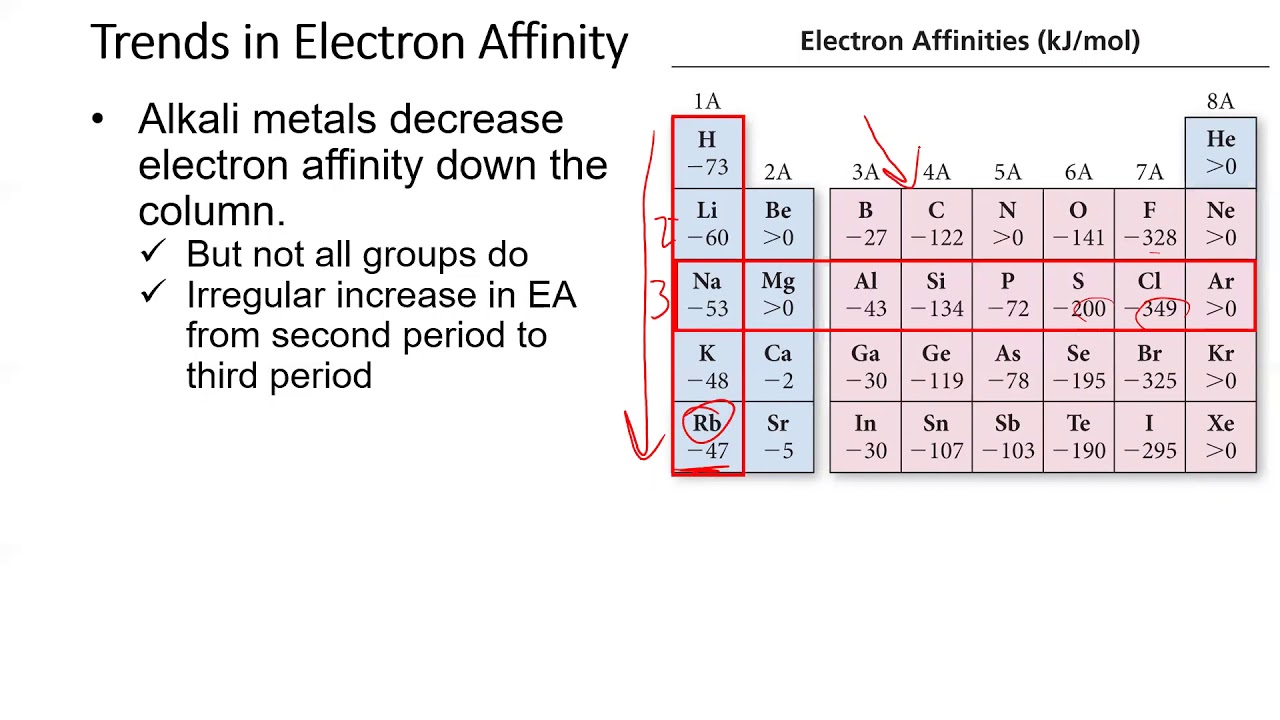
For any reaction that releases energy, the change Δ E in total energy has a negative value and the reaction is called an exothermic process. 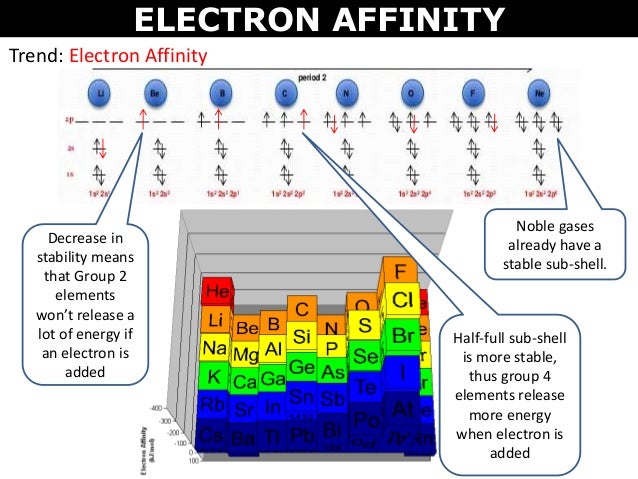
To use electron affinities properly, it is essential to keep track of sign. Together they may undergo charge-transfer reactions. Another example, a molecule or atom that has a more positive value of electron affinity than another is often called an electron acceptor and the less positive an electron donor. Other theoretical concepts that use electron affinity include electronic chemical potential and chemical hardness. Mulliken to develop an electronegativity scale for atoms, equal to the average of the electronsĪffinity and ionization potential. This property is used to measure atoms and molecules in the gaseous state only, since in a solid or liquid state their energy levels would be changed by contact with other atoms or molecules.Ī list of the electron affinities was used by Robert S. Measurement and use of electron affinity 4 "Electron affinity" as defined in solid state physics.1 Measurement and use of electron affinity.Additional calculations on methane C-H bond addition to 1-X afford reaction and activation energy trends that correlate with the reaction energies of H2 addition leading to the Y-product. 
Thus, different complexes such as 1-O and 1-CH2 can have very similar reaction energies for H2 addition arising from opposing hydride and proton affinity effects. The analysis highlights the importance of the electron and hydride affinities, which are not commonly used in rationalizing trends of oxidative addition reactions. To better understand the origin of the computed trends, the thermodynamics of H2 addition are analyzed using a cycle of hydride and proton addition steps. 
No simple correlations are found between the computed reaction energies of H2 addition and either the IEs or EAs. One electronic state has a d(6)-(a)(2)(b)(2)(b)(2) configuration and a square pyramidal geometry, the other a d(6)-(a)(2)(b)(2)(a)(2) configuration with a distorted-Y trigonal bipyramidal geometry. Two closed shell singlet states differentiated in the C2 point group by the d(6)-electon configuration are investigated for the five-coordinate Ir(III) dihydride product. The calculations are also used to study the effects of varying X on the thermodynamics of oxidative H2 addition to 1-X. The results exhibit complex trends, but can be satisfactorily accounted for by invoking a combination of electronegativity and specific π-orbital effects. Electron affinity trend free#Two distinct σ-type ((2)A1 or (2)A) and π-type ((2)B1 or (2)B) electronic states are calculated for each of the free radical cation and anion. Both C2v and C2 geometries are considered. Electron affinity trend series#DFT methods are used to calculate the ionization energy (IE) and electron affinity (EA) trends in a series of pincer ligated d(8)-Ir((tBu4)PXCXP) complexes (1-X), where C is a 2,6-disubstituted phenyl ring with X = O, NH, CH2, BH, S, PH, SiH2, and GeH2. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
